

依据实验组前期对薄膜沉积的实验,选择-0.4mA进行两电极的恒流沉积,并用椭偏仪进行在位监测,每沉积180s后进行300nm到800nm的椭偏测试。即在沉积180s、360s、540s、720s、900s、1080s后分别进行了椭偏仪全谱测试,测试角度为70°。
展示全部 
椭偏仪在位表征电化学沉积的系统搭建(二十二)- 沉积前装置的椭偏数据
1形貌分析
图4-2(a)是准在位监测后沉积1080s时测试得到的SEM图,在1μm放大倍数下看到沉积的薄膜颗粒大小不等(~10-103nm),形态上为不规则的块状。实验组前期用三电极体系(Au/Si工作电极,Pt网对电极,Ag/AgCl参比电极)恒压法(-0.05V)常温下(T=20℃)沉积30分钟得到的结果如图4-2(b)所示。与恒压沉积相比,沉积薄膜粒径不均匀性更强。

图4-2CU20薄膜的SEM图:(a)沉积1080s(b)实验组前期恒压常温沉积
2不同沉积时间椭偏数据的分析
对沉积时间为180s、360s、540s、720s、900s、1080s的CU20薄膜分别进行了椭偏仪全谱(300-800nm)测试,获得椭偏参数以及样品整体的反射率和光学常数。
2.1沉积前装置的椭偏数据
1、Psi、Delta、α、R实验数据在-0.4mA时进行两电极的恒流沉积,并在每沉积180s以后进行300nm到800nm的椭偏测试。在这个沉积电压下,不同时间沉积的库仑量如表4-1所示。由X-ray测试知该电流下沉积的为CU20,假设法拉第效率为100%,则所对应的生长速率分别为0.94nm/s,相应沉积时间下的CU20沉积厚度如表4-1所示。
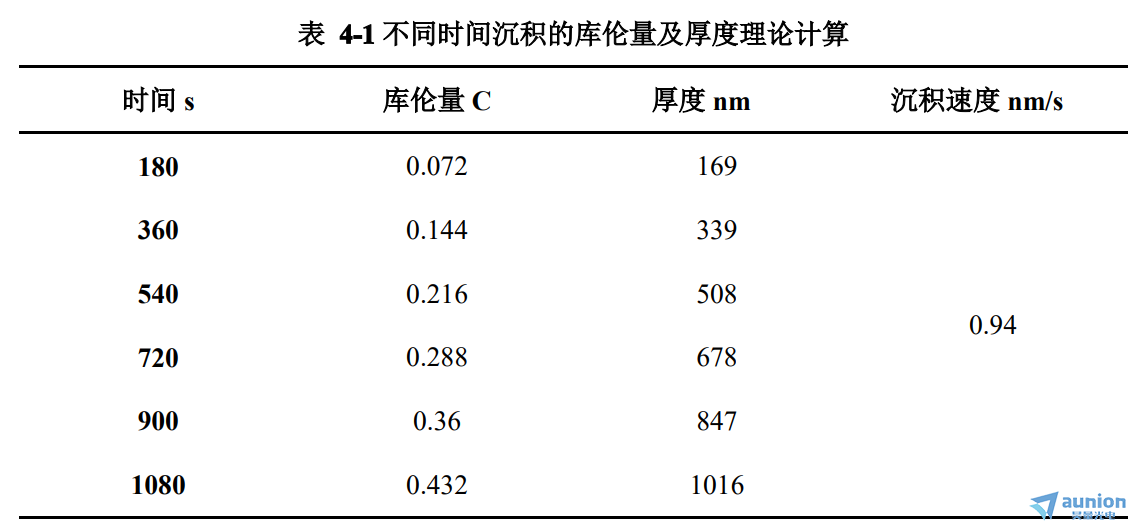
如图4-3(a)所示,在沉积之前测试的Psi和Delta随波长增加而增加,在500nm处有跃迁,在长波段(600nm-800nm)存在波动。图4-3(b)是对应的吸收系数α和反射率R值随波长的变化图,可以看到R值在500nm处存在跃迁,趋近于zui大值1后,经文献查阅知这属于基底Au的反射特性。说明没有沉积之前所得到的椭偏测试结果主要反应的是衬底的信息,ITO和溶液对其影响甚小,也进一步证明该流动型装置用于监测薄膜沉积是可行的。对于α值,在370nm和600nm附近存在吸收峰,其和文献中报道的ITO玻璃基板上Au纳米膜的连续可见光吸收光谱出现的峰位十分接近,相对于文献其峰位发生蓝移且两峰值存在差异,这可能是由于Au薄膜上溶液和ITO带来的影响。

图4-3 沉积0s时(a)Psi和Delta(b)R随波长变化
2.2装置对应的光学常数
图4-4(a)是沉积之前测试得到的n、k随波长的变化图,从图中可以看到短波段图线较平滑,长波段数据波动大。n值在500nm附近出现峰,k值在600nm附近出现峰。500nm处n值存在跃迁,说明该处附近可能有等离子体共振峰的出现。
图4-4(b)是沉积之前测试得到的 、
、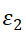 ,从图中可以看到短波段数据曲线平滑,长波段数据波动大。
,从图中可以看到短波段数据曲线平滑,长波段数据波动大。 、
、 均在500nm附近出现峰,这归因于Au表面等离子体共振。
均在500nm附近出现峰,这归因于Au表面等离子体共振。

图4-4 沉积0s时的n、k、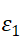 、
、 随波长的变化
随波长的变化
经过以上分析可知,在该体系下(ITO-溶液-Au/Si),较短波段得到的椭偏参数比较光滑,在较长波段得到椭偏数据波动比较大。与较短波段相比长波段得到的数据误差更小,该测试系统更适合较短波段测试。
了解更多椭偏仪详情,请访问上海昊量光电的官方网页:
https://www.auniontech.com/three-level-56.html
更多详情请联系昊量光电/欢迎直接联系昊量光电
关于昊量光电:
上海昊量光电设备有限公司是光电产品专业代理商,产品包括各类激光器、光电调制器、光学测量设备、光学元件等,涉及应用涵盖了材料加工、光通讯、生物医疗、科学研究、国防、量子光学、生物显微、物联传感、激光制造等;可为客户提供完整的设备安装,培训,硬件开发,软件开发,系统集成等服务。
您可以通过我们昊量光电的官方网站www.auniontech.com了解更多的产品信息,或直接来电咨询4006-888-532。
参考文献
[1] WONG H S P, FRANK D J, SOLOMON P M et al. Nanoscale cmos[J]. Proceedings of the IEEE, 1999, 87(4): 537-570.
[2] LOSURDO M, HINGERL K. ellipsometry at the nanoscale[M]. Springer Heidelberg New York Dordrecht London. 2013.
[3] DYRE J C. Universal low-temperature ac conductivity of macroscopically disordered nonmetals[J]. Physical Review B, 1993, 48(17): 12511-12526. DOI:10.1103/PhysRevB.48.12511.
[4] CHEN S, KÜHNE P, STANISHEV V et al. On the anomalous optical conductivity dISPersion of electrically conducting polymers: Ultra-wide spectral range ellipsometry combined with a Drude-Lorentz model[J]. Journal of Materials Chemistry C, 2019, 7(15): 4350-4362.
[5] 陈篮,周岩. 膜厚度测量的椭偏仪法原理分析[J]. 大学物理实验, 1999, 12(3): 10-13.
[6] ZAPIEN J A, COLLINS R W, MESSIER R. Multichannel ellipsometer for real time spectroscopy of thin film deposition from 1.5 to 6.5 eV[J]. Review of Scientific Instruments, 2000, 71(9): 3451-3460.
[7] DULTSEV F N, KOLOSOVSKY E A. Application of ellipsometry to control the plasmachemical synthesis of thin TiONx layers[J]. Advances in Condensed Matter Physics, 2015, 2015: 1-8.
[8] DULTSEV F N, KOLOSOVSKY E A. Application of ellipsometry to control the plasmachemical synthesis of thin TiONx layers[J]. Advances in Condensed Matter Physics, 2015, 2015: 1-8.
[9] YUAN M, YUAN L, HU Z et al. In Situ Spectroscopic Ellipsometry for Thermochromic CsPbI3 Phase Evolution Portfolio[J]. Journal of Physical Chemistry C, 2020, 124(14): 8008-8014.
[10] 焦杨景.椭偏仪在位表征电化学沉积的系统搭建.云南大学说是论文,2022.
[11] CANEPA M, MAIDECCHI G, TOCCAFONDI C et al. Spectroscopic ellipsometry of self assembLED monolayers: Interface effects. the case of phenyl selenide SAMs on gold[J]. Physical Chemistry Chemical Physics, 2013, 15(27): 11559-11565. DOI:10.1039/c3cp51304a.
[12] FUJIWARA H, KONDO M, MATSUDA A. Interface-layer formation in microcrystalline Si:H growth on ZnO substrates studied by real-time spectroscopic ellipsometry and infrared spectroscopy[J]. Journal of Applied Physics, 2003, 93(5): 2400-2409.
[13] FUJIWARA H, TOYOSHIMA Y, KONDO M et al. Interface-layer formation mechanism in (formula presented) thin-film growth studied by real-time spectroscopic ellipsometry and infrared spectroscopy[J]. Physical Review B - Condensed Matter and Materials Physics, 1999, 60(19): 13598-13604.
[14] LEE W K, KO J S. Kinetic model for the simulation of hen egg white lysozyme adsorption at solid/water interface[J]. Korean Journal of Chemical Engineering, 2003, 20(3): 549-553.
[15] STAMATAKI K, PAPADAKIS V, EVEREST M A et al. Monitoring adsorption and sedimentation using evanescent-wave cavity ringdown ellipsometry[J]. Applied Optics, 2013, 52(5): 1086-1093.
[16] VIEGAS D, FERNANDES E, QUEIRÓS R et al. Adapting Bobbert-Vlieger model to spectroscopic ellipsometry of gold nanoparticles with bio-organic shells[J]. Biomedical Optics Express, 2017, 8(8): 3538.
[17] ARWIN H. Application of ellipsometry techniques to biological materials[J]. Thin Solid Films, 2011, 519(9): 2589-2592.
[18] ZIMMER A, VEYS-RENAUX D, BROCH L et al. In situ spectroelectrochemical ellipsometry using super continuum white laser: Study of the anodization of magnesium alloy [J]. Journal of Vacuum Science & Technology B, 2019, 37(6): 062911.
[19] ZANGOOIE S, BJORKLUND R, ARWIN H. Water Interaction with Thermally Oxidized Porous Silicon Layers[J]. Journal of The Electrochemical Society, 1997, 144(11): 4027-4035.
[20] KYUNG Y B, LEE S, OH H et al. Determination of the optical functions of various liquids by rotating compensator multichannel spectroscopic ellipsometry[J]. Bulletin of the Korean Chemical Society, 2005, 26(6): 947-951.
[21] OGIEGLO W, VAN DER WERF H, TEMPELMAN K et al. Erratum to ― n-Hexane induced swelling of thin PDMS films under non-equilibrium nanofiltration permeation conditions, resolved by spectroscopic ellipsometry‖ [J. Membr. Sci. 431 (2013), 233-243][J]. Journal of Membrane Science, 2013, 437: 312..
[22] BROCH L, JOHANN L, STEIN N et al. Real time in situ ellipsometric and gravimetric monitoring for electrochemistry experiments[J]. Review of Scientific Instruments, 2007, 78(6).
[23] BISIO F, PRATO M, BARBORINI E et al. Interaction of alkanethiols with nanoporous cluster-assembled Au films[J]. Langmuir, 2011, 27(13): 8371-8376.
[24] 李广立. 氧化亚铜薄膜的制备及其光电性能研究[D]. 西南交通大学, 2016.
[25] 董金矿. 氧化亚铜薄膜的制备及其光催化性能的研究[D]. 安徽建筑大学, 2014.
[26] 张桢. 氧化亚铜薄膜的电化学制备及其光催化和光电性能的研究[D]. 上海交通大学材料科 学与工程学院, 2013.
[27] DISSERTATION M. Cellulose Derivative and Lanthanide Complex Thin Film Cellulose Derivative and Lanthanide Complex Thin Film[J]. 2017.
[28] NIE J, YU X, HU D et al. Preparation and Properties of Cu2O/TiO2 heterojunction Nanocomposite for Rhodamine B Degradation under visible light[J]. ChemistrySelect, 2020, 5(27): 8118-8128.
[29] STRASSER P, GLIECH M, KUEHL S et al. Electrochemical processes on solid shaped nanoparticles with defined facets[J]. Chemical Society Reviews, 2018, 47(3): 715-735.
[30] XU Z, CHEN Y, ZHANG Z et al. Progress of research on underpotential deposition——I. Theory of underpotential deposition[J]. Wuli Huaxue Xuebao/ Acta Physico - Chimica Sinica, 2015, 31(7): 1219-1230.
[31] PANGAROV n. Thermodynamics of electrochemical phase formation and underpotential metal deposition[J]. Electrochimica Acta, 1983, 28(6): 763-775.
[32] KAYASTH S. ELECTRODEPOSITION STUDIES OF RARE EARTHS[J]. Methods in Geochemistry and Geophysics, 1972, 6(C): 5-13.
[33] KONDO T, TAKAKUSAGI S, UOSAKI K. Stability of underpotentially deposited Ag layers on a Au(1 1 1) surface studied by surface X-ray scattering[J]. Electrochemistry Communications, 2009, 11(4): 804-807.
[34] GASPAROTTO L H S, BORISENKO N, BOCCHI N et al. In situ STM investigation of the lithium underpotential deposition on Au(111) in the air- and water-stable ionic liquid 1-butyl-1-methylpyrrolidinium bis(trifluoromethylsulfonyl)amide[J]. Physical Chemistry Chemical Physics, 2009, 11(47): 11140-11145.
[35] SARABIA F J, CLIMENT V, FELIU J M. Underpotential deposition of Nickel on platinum single crystal electrodes[J]. Journal of Electroanalytical Chemistry, 2018, 819(V): 391-400.
[36] BARD A J, FAULKNER L R, SWAIN E et al. Fundamentals and Applications[M]. John Wiley & Sons, Inc, 2001.
[37] SCHWEINER F, MAIN J, FELDMAIER M et al. Impact of the valence band structure of Cu2O on excitonic spectra[J]. Physical Review B, 2016, 93(19): 1-16.
[38] XIONG L, HUANG S, YANG X et al. P-Type and n-type Cu2O semiconductor thin films: Controllable preparation by simple solvothermal method and photoelectrochemical properties[J]. Electrochimica Acta, 2011, 56(6): 2735-2739.
[39] KAZIMIERCZUK T, FRÖHLICH D, SCHEEL S et al. Giant Rydberg excitons in the copper oxide Cu2O[J]. Nature, 2014, 514(7522): 343-347.
[40] RAEBIGER H, LANY S, ZUNGER A. Origins of the p-type nature and cation deficiency in Cu2 O and related materials[J]. Physical Review B - Condensed Matter and Materials Physics, 2007, 76(4): 1-5.
[41] 舒云. Cu2O薄膜的电化学制备及其光电化学性能的研究[D]. 云南大学物理与天文学院,2019.
展示全部 